Community Engagement
At Fate, we believe that supporting the communities where we live is just as important as the work we do. We contribute to our local community through employee volunteer opportunities and financial support of local organizations.
We Are Building a Highly Qualified and Diverse, Equitable and Inclusive Workforce
We believe all employees should bring their whole selves to work, where they can challenge the status quo and use their intellectual curiosity to bring innovative medicines to those suffering from autoimmune diseases and cancer.
Our commitment to diversifying the representation of our organization is a commitment that starts at the top.
Fate’s Workforce as of December 31, 2024
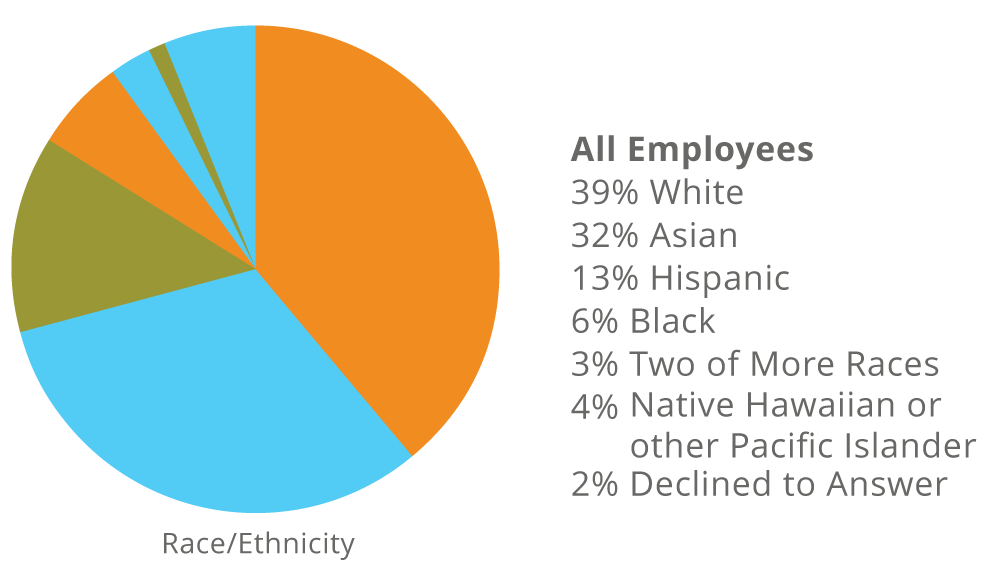
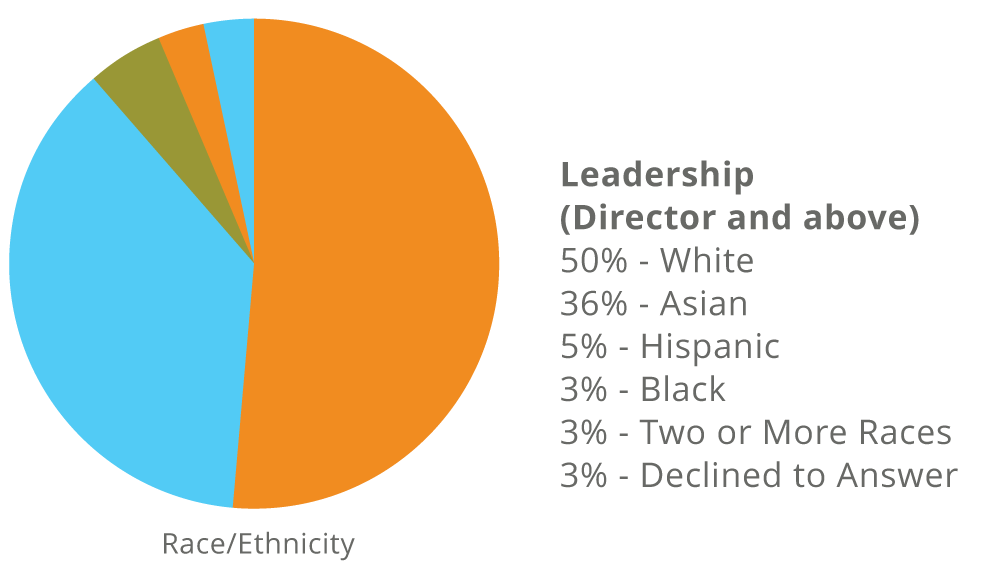
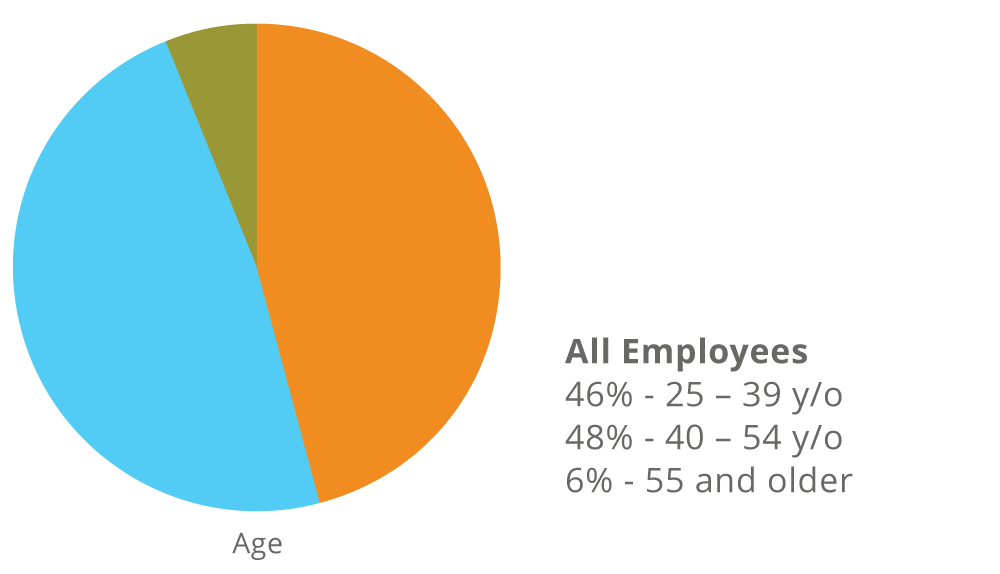
Women make up 56% of our entire workforce and 56% of our leadership (Director and above).
*Employee race/ethnicity and gender are self-disclosed on a voluntary basis.
Employee Engagement
At Fate, we believe investing in our employees’ personal and professional growth and wellbeing makes us a stronger company. To learn about how we engage, support and develop our employees, visit our Careers & Culture pages.
Employee Health & Safety
Our employees are our most valuable assets, and their safety and health are our priority. We are committed to creating a workplace that is safe, healthy, and injury-free. We employ personnel dedicated to ensuring that proactive health, safety, and environmental management programs are maintained throughout our organization and are prioritized as part of our corporate philosophy.
As part of our commitment to employee safety and environmental compliance we periodically conduct gap analyses focused on EH&S sustainability at all levels of our organization. We have several cross-functional committees to support our EH&S sustainability, including an EH&S Committee, a Biological Safety Committee, and a Business Continuity Management Committee.
Employee involvement is vital to sustaining a safe and compliant work environment.
We mandate a comprehensive role-based employee training program that all employees must participate in as part of their annual performance goals. Training topics include bloodborne pathogens, injury illness prevention, hazardous waste management/recycling, emergency action plan, and security management.
We also maintain an Area Safety Representative (ASR) program to enhance communication and employee participation around EH&S matters. ASRs are trained by our EH&S department to perform routine inspections and also receive specialized training such as First Aid, CPR, and emergency spill response. The ASR program is a volunteer-based program and we typically have ~10 employees designated as ASRs throughout the organization at a given time.
Environmental Sustainability
We are committed to operating our facilities in an environmentally responsible way to reduce our environmental impact and protect our staff, our business, and the communities where we operate.
Prior to moving into our current corporate headquarters in 2021, we undertook extensive tenant improvements to the facility. As part of that project, we successfully diverted 482 tons of material from landfill to recycling. This made up 57% of all material removed during the demolition phase and waste produced during the course of construction.

Our corporate headquarters have been designed with the following features:
- Energy efficient air handling units, boilers, LED lighting, and other energy intensive items installed throughout the facilities to reduce energy consumption.
- Our facilities are outfitted with Smart Building solutions, such as occupancy sensors, lighting controls, automatic shade controls, and air conditioning units reducing airflow based on occupancy.
- A fully integrated Building Management System (BMS) was installed and we employ personnel dedicated to monitoring the BMS to ensure that our building environmental conditions are being maintained in an energy efficient manner at all times.
- Electric Vehicle car charging stations are provided free of charge to employees, to encourage the use of electric vehicles.
- Water-saving bathroom faucets and toilets are installed throughout all our facilities. Water consumption is closely monitored, and we continue to work to reduce it where possible.
- Drought tolerant landscaping around our facilities dramatically reduces the volume of water utilized to maintain the plant life around the building.
- In 2024 we implemented a lab recycling program diverting 50% of lab generated waste from landfills and designed to lower our environmental footprint.
We continue to be mindful of how we can minimize our environmental footprint in our research and manufacturing operations.
Clinical Trial Standards
Although we do not currently commercialize or market any prescription cellular immunotherapy products, we do have several clinical trials ongoing for our product candidates, and we are committed to conducting our clinical trials ethically and adhering to best practice principles. We follow the International Council on Harmonisation’s Good Clinical Practices (ICH GCP), an international ethical and scientific quality standard for designing, conducting, recording and reporting trials that involve the participation of human subjects. We also require our contract research organizations and other third parties we work with to follow ICH GCP, all applicable regulations in the countries where the clinical study is being performed, and the trial protocol.
Prior to commencing any clinical trial, we submit an Investigational New Drug (IND) application to the US Food and Drug Administration (FDA). Additionally, each clinical trial site must receive approval for the clinical trial from an independent Institutional Review Board (IRB) before commencing trial activities at that site. Once a clinical trial commences, we take responsibility for ensuring that the clinical trial is conducted in accordance with the investigational plan outlined in the trial protocol, ICH GCP and all applicable regulations, the rights and well-being of the subjects are protected, and the study data is accurate and reliable.
Drug Quality & Safety
Patient safety is a company-wide imperative at Fate. We are committed to the continuous evaluation of the benefits and risks of our investigational immunotherapies and proactively take action to protect the safety and well-being of our patients.
We have policies in place that uphold regulatory standards, taking a proactive approach to ensure compliance and performing routine compliance checks. We also have a dedicated drug safety team to review and evaluate the safety of our medicines.

Our investigational immunotherapies are subject to extensive regulations focused on quality and safety:
- All investigational immunotherapies must be manufactured in accordance with current good manufacturing practices (cGMP) requirements, using qualified facilities, equipment, and materials, with oversight by our Quality Assurance unit to assure appropriate cGMP manufacturing and product release.
- Our internal Safety Assessment Committee manages the safety of our investigational immunotherapies and communication of safety matters with key stakeholders such as regulators, clinical investigators, and clinical sites’ Institutional Review Boards.
- From time to time we engage an independent group of qualified experts, known as an Independent Data Monitoring Committee (IDMC), to provide an independent review of clinical trial data at designated checkpoints and advise us regarding the continuing safety of clinical trial subjects. Through its regular review of data, the IDMC may recommend changes to the trial design based on emerging safety concerns, including termination of the trial if it identifies an unacceptable safety risk for subjects.
Ethics & Compliance
We are committed to conducting our business ethically and operating with integrity. Our Code of Business Conduct and Ethics (Code of Conduct) governs the behavior of all Fate directors, officers and employees and reinforces the ethical and compliance principles that guide our daily operations. We provide our Code of Conduct to all employees and require each employee to sign periodic certifications confirming they have reviewed the Code of Conduct and the requirements set forth therein.
If an employee witnesses a violation of the Code of Conduct, or any law or regulation, they are required to report it immediately. Employees may choose to report their concerns anonymously to our Compliance Officer or directly to our Audit Committee. Fate maintains a strict policy against any form of retaliation for an employee reporting in good faith a suspected Code of Conduct violation.
A copy of our Code of Conduct can be found here »
Governance
Our Board of Directors plays a valuable role in the direction and oversight of our business. Our Board is comprised of ten individuals with strong backgrounds in executive leadership and management, accounting and finance, and industry knowledge. Collectively, our Board possesses a deep and broad set of skills and experience.
40%
of our board is female
30%
of our board is a racial or ethnic minority
We have three standing committees which oversee our main governance functions: the Audit Committee, the Compensation Committee and Nominating and Corporate Governance Committee. Each board committee meets regularly and reports to the full Board of Directors from time to time and whenever requested to do so by the Board.

For additional information on the committees, see our committee charters:
Our board also has several advisory committees.
We are committed to independent leadership on our board. Our CEO does not serve as the Board Chairman and nine of our ten Board members are independent. Our three standing committees are composed entirely of independent directors in accordance with NASDAQ listing standards.
Cybersecurity
We are committed to maintaining a secure information technology environment. The Audit Committee of our Board of Directors has primary oversight and responsibility for cybersecurity risk and provides regular reports to our Board on our cybersecurity program. In addition, our Board reviews our enterprise risk management program on at least an annual basis. We have a dedicated team of cybersecurity professionals focused on protecting the confidentiality, integrity, and availability of the personal and corporate information we manage. We provide ongoing education to our employees on potential threats, including a phishing awareness training program.
We also conduct regular penetration testing and security audits by third-party experts to ensure that our cybersecurity defense is up to date and equipped to combat the latest methods used by hackers and bad agents.
Health and Wellbeing
At Fate we believe that wellness includes many areas of life that go beyond an individual’s physical health, including mental and financial wellness. We maintain a state-of-the art fitness center on-site at our headquarters.
- Employees have access to an onsite Fitness Center with sports courts and an on-demand fitness app. Healthy food options are also available to purchase on-site.
- Regular webinars are held on financial wellbeing, in addition to several on demand tools, resources and trainings.

Compensation and Benefits
At Fate, we believe our employees should receive equal pay for equal work, regardless of their gender, race, or other protected characteristics, and we comply with all relevant state equal pay laws. To deliver on that commitment to our employees and our stakeholders, we benchmark and set pay ranges based on relevant market data, also considering factors such as an employee’s role/duties, experience, skill level, and job location. We regularly review our compensation practices, both in terms of our overall workforce and individual employees, to strive for pay that is fair and equitable. In 2022, we worked with an independent third-party to conduct a company-wide pay equity analysis. The analysis focused on identifying statistically significant inequities based on gender, age, and ethnicity. We were pleased to confirm that there were no significant deficiencies or disparities identified and the company’s realized pay practices result in pay consistent with our compensation philosophy. We continue to regularly review and analyze our pay and compensation practices so that we can proactively take measures to contribute to equitable pay for all our workforce.
For more information about the compensation and benefits we offer our employees, visit our Careers & Culture page.



